GST-Tag (1-224) Schistosoma Protein
CAT#: AR03011PU-N
GST-Tag (1-224) schistosoma recombinant protein, 0.1 mg
|
Need it in bulk or customized? Get a free quote |
CNY 8,900.00
货期*
详询
规格
Specifications
| Product Data | |
| Species | Schistosoma |
| Expression Host | E. coli |
| Expression cDNA Clone or AA Sequence |
MSPILGYWKI KGLVQPTRLL LEYLEEKYEE HLYERDEGDK WRNKKFELGL EFPNLPYYID GDVKLTQSMA IIRYIADKHN MLGGCPKERA EISMLEGAVL DIRYGVSRIA YSKDFETLKV DFLSKLPEML KMFEDRLCHK TYLNGDHVTH PDFMLYDALD VVLYMDPMCL DAFPKLVCFK KRIEAIPQID KYLKSSKYIA WPLQGWQATF GGGDHPPKSD LVPR
|
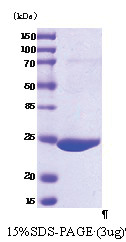
| Predicted MW | 26 kDa |
| Concentration | lot specific |
| Purity | >95% by SDS PAGE |
| Buffer | Presentation State: Purified State: Liquid purified protein Buffer System: Phosphate Buffered Saline pH 7.4 |
| Preparation | Liquid purified protein |
| Protein Description | Recombinant GST was expressed in E.coli and purified by conventional chromatography techniques. |
| Storage | Store undiluted at 2-8°C for one month or (in aliquots) at -20°C to -80°C for longer. Avoid repeated freezing and thawing. |
| Stability | Shelf life: one year from despatch. |
| Bioactivity | Biological: 0.5-2.5 units/mg (please enquire for specific batch value). A unit is defined as the amount of enzyme that conjugate 1.0 u mole of 1-chloro-2,4-dinitrobenzene (CDNB) with reduced glutathione per minute at pH 6.5 at 25°C. Activity Assay 1. Prepare a 1 ml reaction mix into a suitable container: The final concentrations are 97 mM potassium phosphate, 0.97 mM EDTA, 2.5 mM glutathione, reduced, 1.0 mM 1-chloro-2,4-dinitrobenzene (CDNB), 3.2% (v/v) ethanol. 2. Equilibrate to 25°C and monitor at A340nm until the value is constant using a spectrophotometer. 3. Add 50 ul of GST protein with various concentrations (1ug, 2ug, 5ug) in 950 ul reaction buffer. 4. Mix by inversion and record the increase at A340nm for 5 minutes. |
| Endotoxin | < 1.0 EU per 1 ug of protein (determined by LAL method) |
| Reference Data | |
| Summary | Glutathione S-transferase (GST) represents a major group of detoxification enzymes. This enzyme acts by catalyzing the reaction of glutathione with an acceptor molecule to form an S-substituted glutathione (S=sulfur). The reactions utilizing glutathione contribute the transformation of a wide range of compounds, including carcinogens, therapeutic drugs, and products of oxidative stress. As well as its enzymatic activities, GST may also bind toxins and function as transport protein. Because of this, an early term for GSTs was ligandin. Glutathione S-transferase was originally separated from Schistosoma japonicum but currently isolated from recombinant E.coli source. |
Documents
| FAQs |
| SDS |
Customer
Reviews
Loading...


 United States
United States
 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China