Thioredoxin-1 (TRX1) (2-109, His-tag) Escherichia coli Protein
CAT#: AR09459PU-L
Thioredoxin-1 (TRX1) (2-109, His-tag) e. coli recombinant protein, 0.25 mg
|
Need it in bulk or customized? Get a free quote |
CNY 14,920.00
货期*
详询
规格
| Cited in 1 publication. |
Specifications
| Product Data | |
| Species | Escherichia coli |
| Expression Host | E. coli |
| Expression cDNA Clone or AA Sequence |
MHHHHHHMGS DKIIHLTDDS FDTDVLKADG AILVDFWAEW CGPCKMIAPI LDEIADEYQG KLTVAKLNID QNPGTAPKYG IRGIPTLLLF KNGEVAATKV GALSKGQLKE FLDANLA
|
| Tag | His-tag |
| Predicted MW | 12.8 kDa |
| Concentration | lot specific |
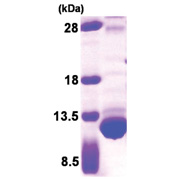
| Purity | >85% by SDS – PAGE |
| Buffer | Presentation State: Purified State: Liquid purified protein Buffer System: 20 mM Tris-HCl buffer (pH 8.0) containing 10% glycerol, 1 mM DTT |
| Preparation | Liquid purified protein |
| Protein Description | Recombinant E.coli TXN protein, fused to His-tag at N-terminus, was expressed in E.coli and purified by using conventional chromatography techniques. |
| Storage | Store undiluted at 2-8°C for up to two weeks or (in aliquots) at -20°C or -70°C for longer. Avoid repeated freezing and thawing. |
| Stability | Shelf life: one year from despatch. |
| Bioactivity | Biological: Specific activity is >70 A650/cm/min/mg, obtained by measuring the increase of insulin precipitation in absorbance at 650 nm resulting from the reduction of insulin. |
| Reference Data | |
| Summary | Thioredoxin 1 (Trx) is a low molecular weight redox protein. Trx contains a redox active disulfide/dithiol group within the conserved Cys-Gly-Pro-Cys active site. It is involved in the first unique step in DNA synthesis. Trx also provides control over a number of transcription factors affecting cell proliferation and death through a mechanism referred to as redox regulation. |
Citations (1)
| The use of this Proteins has been cited in the following citations: |
|---|
|
Baicalein exhibits anti-inflammatory effects via inhibition of NF-κB transactivation.
,null,
Biochemical pharmacology
,PubMed ID 27019135
|
Documents
| FAQs |
| SDS |
Customer
Reviews
Loading...


 United States
United States
 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China