GCLM / GLCLR (1-274, His-tag) Human Protein
CAT#: AR50481PU-S
GCLM / GLCLR (1-274, His-tag) human recombinant protein, 0.1 mg
Size: 500 ug
|
Need it in bulk or customized? Get a free quote |
CNY 8,900.00
货期*
详询
规格
Specifications
| Product Data | |
| Species | Human |
| Expression Host | E. coli |
| Expression cDNA Clone or AA Sequence |
MGSSHHHHHH SSGLVPRGSH MGSHMGTDSR AAKALLARAR TLHLQTGNLL NWGRLRKKCP STHSEELHDC IQKTLNEWSS QINPDLVREF PDVLECTVSH AVEKINPDER EEMKVSAKLF IVESNSSSST RSAVDMACSV LGVAQLDSVI IASPPIEDGV NLSLEHLQPY WEELENLVQS KKIVAIGTSD LDKTQLEQLY QWAQVKPNSN QVNLASCCVM PPDLTAFAKQ FDIQLLTHND PKELLSEASF QEALQESIPD IQAHEWVPLW LLRYSVIVKS RGIIKSKGYI LQAKRRGS
|
| Tag | His-tag |
| Predicted MW | 33.3 kDa |
| Concentration | lot specific |
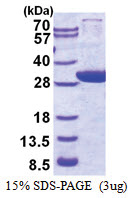
| Purity | >90% by SDS - PAGE |
| Buffer | Presentation State: Purified State: Liquid purified protein Buffer System: 20 mM Tris-HCl buffer (pH 8.0) containing 10% glycerol, 0.15M NaCl |
| Preparation | Liquid purified protein |
| Protein Description | Recombinant human GCLM protein, fused to His-tag at N-terminus, was expressed in E.coli and purified by using conventional chromatography techniques. |
| Storage | Store undiluted at 2-8°C for one week or (in aliquots) at -20°C to -80°C for longer. Avoid repeated freezing and thawing. |
| Stability | Shelf life: one year from despatch. |
| Reference Data | |
| RefSeq | NP_001295182 |
| Locus ID | 2730 |
| UniProt ID | P48507 |
| Cytogenetics | 1p22.1 |
| Synonyms | GLCLR |
| Summary | Glutamate-cysteine ligase, also known as gamma-glutamylcysteine synthetase, is the first rate limiting enzyme of glutathione synthesis. The enzyme consists of two subunits, a heavy catalytic subunit and a light regulatory subunit. Gamma glutamylcysteine synthetase deficiency has been implicated in some forms of hemolytic anemia. Alternative splicing results in multiple transcript variants encoding different isoforms. [provided by RefSeq, Apr 2015] |
| Protein Families | Druggable Genome |
| Protein Pathways | Glutathione metabolism, Metabolic pathways |
Documents
| FAQs |
| SDS |
Customer
Reviews
Loading...


 United States
United States
 Germany
Germany
 Japan
Japan
 United Kingdom
United Kingdom
 China
China